Background
MAX phases are ternary carbides and nitrides with unique combinations of ceramic properties (e.g., resistance to high temperature, fatigue, and damage) and metallic properties (e.g., high electrical and thermal conductivity). They can be used, for example, as coatings for electronics operating under extreme conditions. Synthesis of MAX phases is typically accomplished by traditional solid-state methods, such as hot isostatic pressing, spark plasma sintering, or simple high-temperature furnace techniques. While these methods are well established, the final product's shape is difficult to control because the use of elemental or binary carbides/nitrides/intermetallics as reactants only allow for powder or pelletized reactant mixtures. On the other hand, the sol-gel approach involves dissolvable metal salts/alkoxides that are transformed and trapped in a gel using a variety of chelating and gel-forming agents. The obtained gel is generally malleable, allowing for shapes otherwise not attainable.
Invention Description
Researchers at Arizona State University have developed processes for producing Cr2GaC MAX phase thick films, microspheres, and hollow microspheres through a sol-gel approach. The synthesis process utilizes the metal-ion-sequestering properties of linear polysaccharides (e.g., chitosan) and crosslinked polysaccharides (e.g., CM-SEPHADEX), and allows the resulting gel to be shaped into micron-sized thick films and microspheres with tunable diameter and porosity. Use of benign and abundant biopolymers is foundational to the simple, convenient, and sustainable nature of this technology.
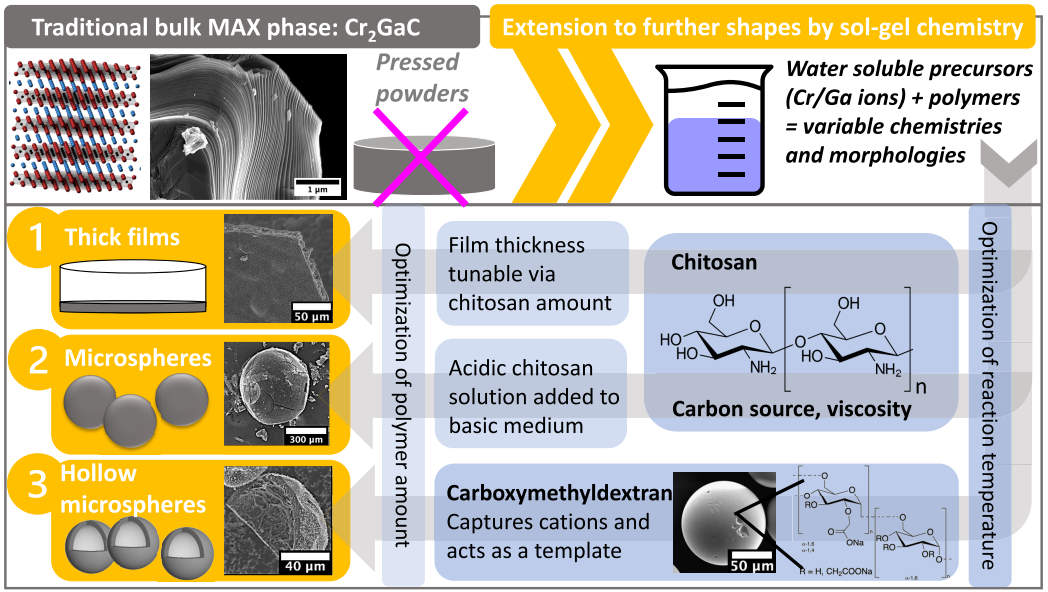
Fig. 1: Schematic illustrating traditional approaches and a more versatile wet chemistry method to achieve a variety of shapes and morphology of MAX phase Cr2GaC.
Potential Applications
• Conductive thick films for micro-electromechanical systems
• Microspheres for fillers and cement additives
• Hollow microspheres for drug delivery, nuclear, and biomedical applications
Benefits and Advantages
• Allows physical properties of MAX phase Cr2GaC to be integrated into thick films and microspheres
• Versatile wet chemistry approach does not require high-end printers or expensive materials
• Easily scalable for large-volume, potentially continuous production
Related Publication: Shape Control of MAX Phases by Biopolymer Sol–Gel Synthesis: Cr2GaC Thick Films, Microspheres, and Hollow Microspheres
Research Homepage of Professor Christina Birkel